
"Sommerfeld formula and Dirac's theory" (PDF). Faced with this grave anomaly, the young Danish physicist Niels Bohr decided to go to England to work together with Rutherford to understand how solar-system. 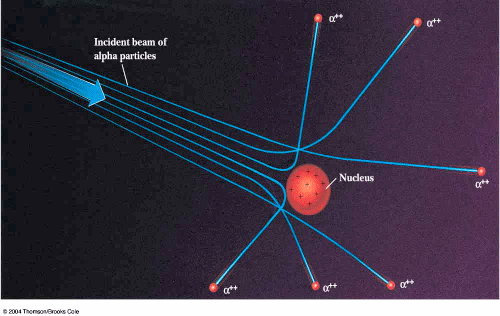
Now click on and select next model, Rutherford’s Classical Solar System model. ^ - Atombau und Spektrallinien, 1921, page 520 Rutherford’s Classical Solar System Model 11."The Kossel-Sommerfeld Theory and the Ring Atom". Based on the above observations, Rutherford proposed the Classical solar system model of atom. Point to Classical solar system model of atom. Here are the limitations of Plum Pudding model. "Einstein's unknown insight and the problem of quantizing chaos" (PDF). Gold foil scattering experiments by Rutherford showed that, Positive charge is not spread evenly over the entire atom. ^ The Collected Papers of Albert Einstein, vol."The quantum theory of radiation and line spectra". Atomic theory is the scientific theory that matter is composed of particles called atoms. Nevertheless, both solutions fail to predict the Lamb shifts. The current theoretical model of the atom involves a dense nucleus surrounded by a probabilistic 'cloud' of electrons. This solution (using substitutions for quantum numbers) is equivalent to the solution of the Dirac equation. The Bohr–Sommerfeld model supplemented the quantized angular momentum condition of the Bohr model with an additional radial quantization condition, the Wilson– Sommerfeld quantization condition ∫ 0 T p r d q r = n h, is the fine-structure constant. Sommerfeld argued that if electronic orbits could be elliptical instead of circular, the energy of the electron would be the same, except in the presence of a magnetic field, introducing what is now known as quantum degeneracy. Bohr–Sommerfeld theory is named after Danish physicist Niels Bohr and German physicist Arnold Sommerfeld. These electrons move around the nucleus in. The Bohr–Sommerfeld model (also known as the Sommerfeld model or Bohr–Sommerfeld theory) was an extension of the Bohr model to allow elliptical orbits of electrons around an atomic nucleus. According to the Rutherford model, the entire mass of the atom is concentrated in a tiny positively charged nucleus. Rutherford designed an experiment using the alpha particles emitted by a radioactive element to probe to the structure of the atom. This simulation gives a microscopic picture of Rutherford's famous experiment in which he shot alpha particles at a thin foil of gold. Rutherford's Classical Solar System Model In 1911, Rutherford overturned Thomson's model with his well-known gold foil experiment where he demonstrated that the atom has a tiny and heavy nucleus. 
The Sommerfeld extensions of the 1913 solar system Bohr model of the hydrogen atom showing the addition of elliptical orbits to explain spectral fine structure. Now you try Plum Pudding to Rutherford's Classical Solar System Model - To answer this question, think about what you observed while doing the Rutherford Scattering simulation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
